agilent.co.jp
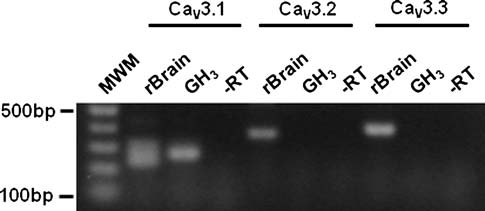
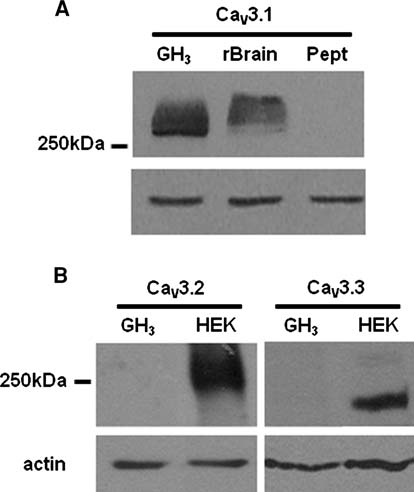
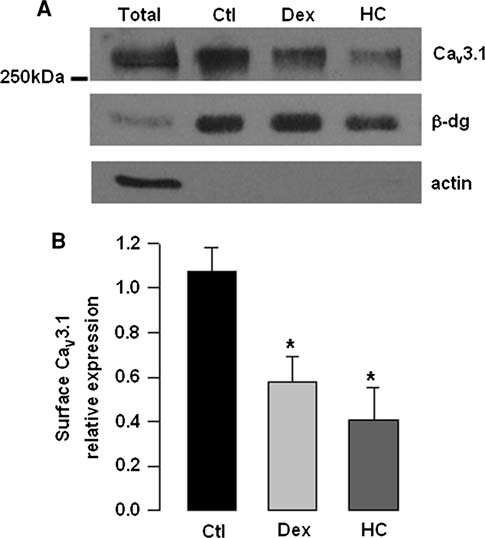
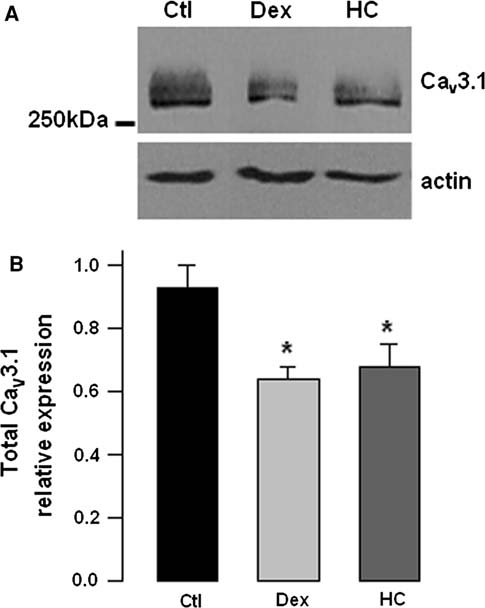
Pharmaceutical Applications Compendium PURITY AND IMPURITY ANALYSIS APPLICATION OF CHROMATOGRAPHY AND MASS SPECTROMETRY IN PHARMACEUTICAL PURITY AND IMPURITY ANALYSIS Accurate assessment of product quality requires robust analytical methods. To remain competitive, drug development and quality control programs must find efficient and effective ways to develop new analytical methods and to transfer and optimize existing procedures. Increased regulatory attention on the control of impurities (e.g., much lower thresholds for genotoxic impurities) has fueled the need for analytical procedures that al ow significantly lower detection limits. This in turn requires more sensitive instruments and places higher demands on selectivity, since many additional impurities may be present at lower concentration ranges. UHPLC offers significant advantages in analytical performance, speed, sensitivity, and resolution. Today's labs must also col aborate with global partners using a variety of instruments. As a result, flexibility in method development and transfer has become a key component of success in the pharmaceutical industry. The application of various mass spectroscopy (MS) detectors to genotoxic impurity analysis has enabled breakthrough detection limits of as little as a few hundred ppm. MS-based methods general y provide additional robustness compared to techniques such as UV alone, due to their high specificity and sensitivity.