Chirurgiadellamanoroma.it

ISSN 0957-4530, Volume 21, Number 6
This article was published in the above mentioned Springer issue.
The material, including all portions thereof, is protected by copyright;
all rights are held exclusively by Springer Science + Business Media.
The material is for personal use only;
commercial use is not permitted.
Unauthorized reproduction, transfer and/or use
may be a violation of criminal as well as civil law.
J Mater Sci: Mater Med (2010) 21:1979–1987 Author's personal copy
In vivo study of ethyl-2-cyanoacrylate applied in direct contactwith nerves regenerating in a novel nerve-guide
A. Merolli • S. Marceddu • L. Rocchi •F. Catalano
Received: 1 January 2010 / Accepted: 22 February 2010 / Published online: 19 March 2010Ó Springer Science+Business Media, LLC 2010
Stitch suture is still the most recommended
method to hold a nerve-guide in place but stitch suture is awell known cause of local inflammatory response. Glues of
The gold standard in treating nerve gap-injuries is the
several kinds have been proposed as an alternative but they
autograft []. Unfortunately, there are several limitations
are not easy to apply in a real surgical setting. In 2006
and complications associated with autografts: (a) harvesting
authors developed a new concept of nerve-guide termed
a donor nerve graft may have significant co-morbidity
‘‘NeuroBox'' which is double-halved, not-degradable and
]; (b) the donor nerve is, often, a smaller sensitive nerve
rigid, and allows the use of cyanoacrylic glues. In this
which limits, from the beginning, a full recovery when a
study, Authors analyzed histologically the nerve-glue
bigger and more important motor nerve requires the treat-
interface. Wistar rats were used as animal model. In group
ment; (c) there is an increasing difficulty in proposing an
1, animals were implanted a NeuroBox to promote the
autograft to patients who neither accept the sacrifice of their
regeneration of an experimentally produced 4 mm gap in
nerves and its associated morbidity nor the lack of a guar-
the sciatic nerve. In group 2, the gap was left without repair
anteed positive outcome (in the worst case, they will per-
(‘‘sham-operated'' group). Group 3 was assembled by
ceive two lesions instead of one); (d) the two stitch sutures
harvesting 10 contralateral intact nerves to document the
securing the autograft (one proximal and one distal) may be
normal anatomy. Semi-thin sections for visible light
the site of an unfavourable fibroblastic proliferation
microscopy and ultra-thin sections for Transmission Elec-
An alternative to autograft is the allograft but it may
tron Microscopy were analyzed. Results showed that
bring the even greater problem of a life-long immunosup-
application of ethyl-2-cyanoacrylate directly to the epi-
pressive therapy []. A new class of commercial allografts
neurium produced no significative insult to the underlining
are now commercially available as non-immunogenic to
nerve fibers nor impaired nerve regeneration. No regener-
the host, since they are so highly processed that only the
ation occurred in the ‘‘sham-operated'' group.
Laminin-laden structure of the original nerve fasciclesremains []; this treatment assimilate them to multichan-neled artificial nerve guides.
Artificial nerve guides (or conduits) have been intro-
duced into clinical practice more than 20 years ago; theyare cylindrical conduits inside which a regenerating nervestump may find protection and guidance An overview
A. Merolli (&) � L. Rocchi � F. Catalano
of the clinical outcome showed that they perform at least as
Orthopaedics and Hand Surgery Unit, The Catholic University
good as autografts in peripheral nerve injuries where gaps
School of Medicine in Rome, Complesso ‘‘Columbus'',via Moscati 31, 00168 Rome, Italy
are not-longer-than 20 mm; in this situation they bring the
advantage of avoiding donor site sacrifice and morbidityand provide an easier and quicker surgical technique ,
Nowadays there are several degradable nerve guides in
National Research Council, ISPA-CNR, via dei Mille 48,07100 Sassari, Italy
clinical use ] made of: poly-glycolic-acid (‘‘Neurotube''
J Mater Sci: Mater Med (2010) 21:1979–1987
Author's personal copy
Synovis USA); poly-lactic-acid (‘‘Neurolac'' Ascension
apply in a real surgical setting, where the presence of blood
USA—Polyganics NL); treated bovine collagen (‘‘Neur-
and other fluids is highly variable and little manageable.
agen'' Integra USA; ‘‘Neuroflex'' and ‘‘Neuromatrix''
In 2006 authors developed, and tested in vivo, a new
Stryker—Collagen Matrix USA); a proprietary hydrogel
concept of nerve-guide termed ‘‘NeuroBox'' (patent WO/
non-degradable in vivo (‘‘SaluBridge'' SaluMedica USA).
2008/029373) which is double-halved, not-degradable and
Several other experimental guides have been proposed and
rigid, and does not require the use of any stitch to be
tested in vitro and in vivo [, ].
sutured to the nerve stump, allowing the use of cyanoac-
From the biomaterial point of view, there are three
rylic glues instead. The device proved to allow a successful
structural districts in a nerve guide, namely: (a) the outer
nerve regeneration in vivo [
structure, which is basically the tube inside which the nerve
In this study, Authors analyzed histologically the nerve-
stumps are accommodated; (b) the inner structure, which is
glue interface in vivo. Their hypothesis is that the little
how the tube is filled; (c) the suture, which is the site where
invasiveness in applying ethyl-2-cyanoacrylate directly to
a mechanical force is being applied to the guide and where
the epineurium (which is allowed by the peculiar design of
a mechanical and biological insult is being received by the
the device) is instrumental to the absence of any signifi-
nerve stump.
cative insult to the underlining nerve fibers (which was
Stitch suture is still the most recommended method to
observed) and this contributed significantly to the suc-
hold the guide in place but stitch suture is a well known
cessful nerve regeneration that had taken place.
cause of local inflammatory response (Fig. Gluesof several kinds have been proposed as an alternative[but, unfortunately, they have proved not easy to
2 Materials and methods
Twenty-two male Wistar rats, weighing about 300 g, wereused as animal model. Three groups of samples werestudied. In group 1 (G1), 15 animals were implanted aNeuroBox double-halved stitch-less nerve-guide to pro-mote the regeneration of an experimentally produced 4 mmgap in the sciatic nerve. In group 2 (G2), 7 animals had the4 mm gap left without repair (‘‘sham-operated'' group).
Group 3 (G3) was assembled by harvesting 10 contralateralintact nerves (6 from G1 and 4 from G2) and this group ofsamples documented the normal anatomy of the sciaticnerve in the experimental model.
The NeuroBox was micro-machined from a solid block
of poly-methyl-methacrylate (PMMA) (Repsol, Madrid,Spain) using computer-aided manufacturing (CAM) tech-niques, at the Institute of Bioengineering of Catalunia.
Devices were degreased by sonication in a mix of distilledwater and ethyl alcohol (50–50%) and sterilized by low-temperature
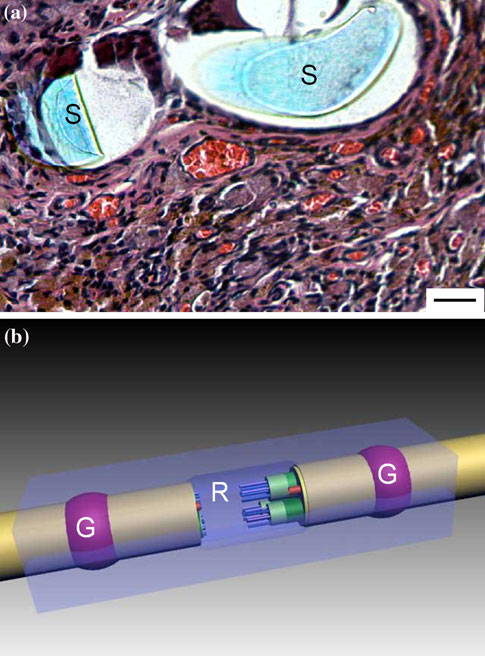
(STERRAD Sterilization Systems, Johnson & Johnson,USA). In the NeuroBox the traditional cylindrical nerveguide is replaced by a box of two-halves into which threemain compartments are recognizable: (1) a lodgement forthe neural stump (one proximal and one distal); (2) acompartment for the acrylic glue (one proximal and onedistal); (3) a flat ‘‘regeneration chamber'', where elongatingaxons from the proximal stump are invited to spread(Fig. b).
Ethyl-2-cyanoacrylate (Loctite, Henkel, Germany) was
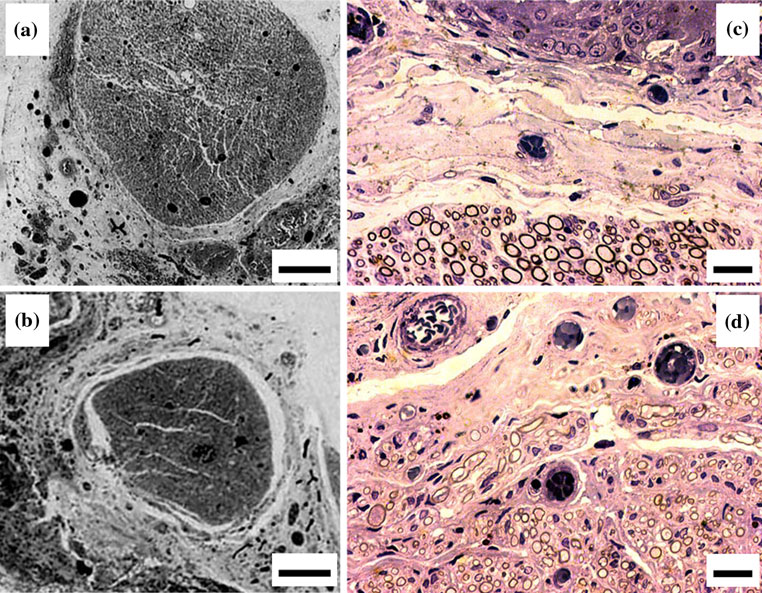
Fig. 1 a A transverse section showing two heads of an epineural knot
employed as cyanoacrylic glue. The dedicated glue-
(S), where a great number of hyperhemic capillaries and hemosiderin
compartment of the NeuroBox promotes the polymeriza-
deposits can be seen (H&E; bar = 10 lm). b Schematic drawing of a
tion of the glue with just the minimum amount which is
NeuroBox device: the volume for the acrylic glue (G) and the flat‘‘regeneration chamber'' (R) are labelled
needed for its wetting. Neural stumps are gently accommodated
J Mater Sci: Mater Med (2010) 21:1979–1987
Author's personal copy
within their compartments, in the bottom half of the guide,
Table 1 Implant and retrieval scheme
just prior the end of the polymerization process. The
acrylic glue conforms to the stumps. The top-half wasgently positioned so to close the device symmetrically
(by wetting, with the glue, the dedicated top-half glue-
compartments). However, the NeuroBox guide cannot be
considered completely sealed but, on the contrary, some
empty space remains at both entrances and fluids and cells
may access the regeneration chamber.
Adequate measures were taken to minimize pain or
Implantation scheme for G1 (NeuroBox), G2 (sham operated group)
discomfort to the animals and experiments were conducted
and G3 (intact contralateral nerve)
in accordance with ECC D86/609 and with the approval ofthe National Committee for Animal Experimentation.
Anaesthesia was induced by 75 mg/kg ketamine chlorhy-
tight. The sciatic nerve was cut proximally to its trifurca-
drate (KETAVET 100, Farmaceutici Gellini, Aprilia, Italy)
tion and a gap of 4 mm in length ensued. Surgical opera-
and 0.5 mg/kg medetomidine chlorhydrate (DOMITOR,
tion required the assistance of optical magnification (Zeiss
Farmos Orion Corporation, Espoo, Finland) with intra-
OP MI 1, Carl Zeiss, Jena, Germany).
muscular injection on the right tight. A 30 mg/kg methyl-
Animals were sacrificed under deep anaesthesia, in an
predinisolone (SOLU-MEDROL, Pharmacia & Up John
atmosphere saturated with CO2; retrievals followed the
NV/SA, Puurs, Belgium) was administered prior to surgery.
timing shown in Table . At retrieval, the nerve is simply
A curvilinear transverse incision with superior convex-
dislodged from the guide. Macroscopic examination of the
ity was performed to gain a smooth access to the intra-
operated site was performed. Retrieved nerves were fixed
muscular interstice to expose the sciatic nerve, in the left
in buffered formaline; post-fixed in osmium tetroxide and
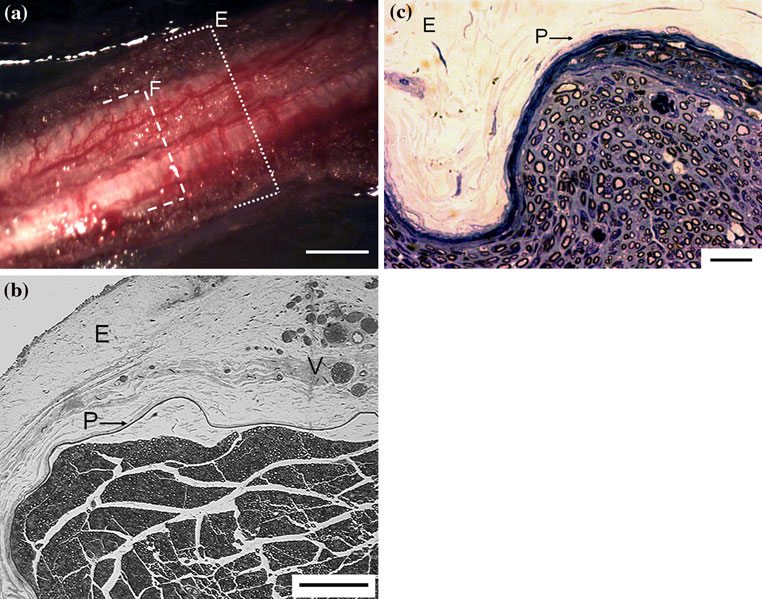
Fig. 2 a Intact sciatic nerve of a Wistar rat shows that the epineurium
(V); a distinct perineurium (P) surrounds the nerve fascicle (toluidine
(E) is a wide bumping protective coat wrapped around the nerve
blue; bar = 100 lm). c Transverse section of an intact sciatic nerve:
fascicles (F), with main vascular trunks and their collateral sinusoidal
the perineurium (P), with its peculiar lamellar arrangement of roughly
branches embedded in (bar = 0.5 mm). b Transverse section of an
7–8 concentric layers, surrounds the nerve fascicle (epineurium is
intact sciatic nerve: the epineurium (E) is made of loose areolar
labelled with E) (H&E; bar = 10 lm)
connective tissue and adipose tissue, with embedded epineural vessels
J Mater Sci: Mater Med (2010) 21:1979–1987
Author's personal copy
dehydrated in serial passages of acetone, then embedded inaraldite. Semi-thin sections were stained for visible lightmicroscopy (Nikon SMZ 800); ultra-thin sections were cutfor Transmission Electron Microscopy (TEM) (Zeiss EM109T). Images were processed by a commercial software(‘‘PhotoDeLuxe'', Adobe).
Five zones were mapped in a retrieved nerve. (1) The
Proximal End (PE), 5 mm proximal to the surgical tran-section. (2) The Proximal Glueing Region (PGR), 1 mm inlength and about 1 mm proximal to the surgical transection(this is the proximal area embraced and locked by thepolymerized acrylate glue). (3) The Regenerate (R). (4)The Distal Glueing Region (DGR), 1 mm in length andabout 1 mm distal to the surgical transection (this is thedistal area embraced and locked by the polymerizedacrylate glue). (5) The Distal End (DE), 5 mm distal to thesurgical transection.
Ethyl-2-cyanoacrylate was applied in direct contact withthe epineural sheath. In the rat, as controls show, Epineu-rium is a wide bumping protective coat wrapped around thenerve fascicle(s) (Fig. a), made of loose areolar connec-tive tissue and adipose tissue, with embedded main vas-cular trunks and their collateral sinusoidal branches(epineural vessels) (Fig. b). A distinct perineurium, with apeculiar lamellar arrangement of roughly 7–8 concentriclayers, surrounds the nerve fibers (Fig. c).
Authors did not find any image of adverse early
inflammatory response or tissue necrosis. No glue appearedinside the epineurium or inside the nerve bundles, in sec-tions taken at the glueing regions or anywhere else, at anytime.
When 6 animals in G1 were sacrificed in an early stage
(2 after 3 days; 2 after 1 week; 2 after 2 weeks; as shownin Table no finding was suggestive of any impairment ofnerve fibers at the level of the glueing regions (Fig. In
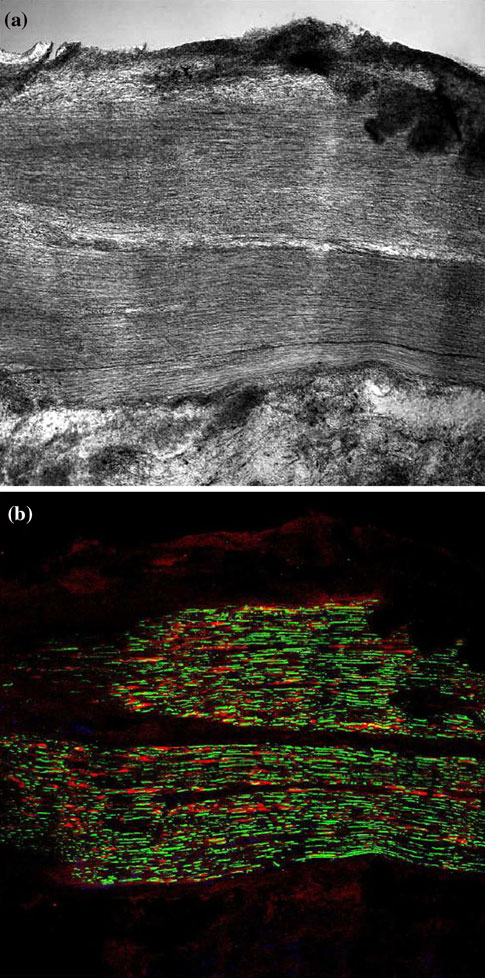
Fig. 3 After 3 days, no morphological alteration of nerve fibers was
this stage, the regenerate consists mostly of an assembled
found at the level of the glueing regions, both in unstained sections
jelly structure of fibrin, blood cells, Schwann's cells and
(a) and in immuno-stained sections (b; green = axons; red =
Schwann's cells) (courtesy of the Institute of Bioengineering of
In G1 nerve regeneration occurred after 1 months and it
was observed in 9/9 cases (3 after 1 month; 3 after2 months; 3 after 3 months; as shown in Table ). The
Epineural sheath which was in direct contact with ethyl-
retrieved nerve was about 4 mm longer than the contra-
2-cyanoacrylate in the proximal and distal glueing regions
lateral intact nerve. There were no signs of any massive
(PGR and DGR) showed no major microscopic alterations
and adverse intraneural fibroblastic proliferation; large and
nor they were found in the underlining nerve fibers; a clear
small myelinated fibers were identified and also several
fascicular structure, well demarcated from the surrounding
non-myelinated axons; fine blood vessels were well rep-
epineurium, was preserved (Fig. ).
resented (confirming what already reported ]). A fibrous
More in details, in the PGR the normal structure of
capsule was found around the guide but not inside the
the nerve was preserved, with myelinated large diameter
regeneration chamber (Fig.
fibers and smaller non-myelinated fibers and intraneural
J Mater Sci: Mater Med (2010) 21:1979–1987
Author's personal copy
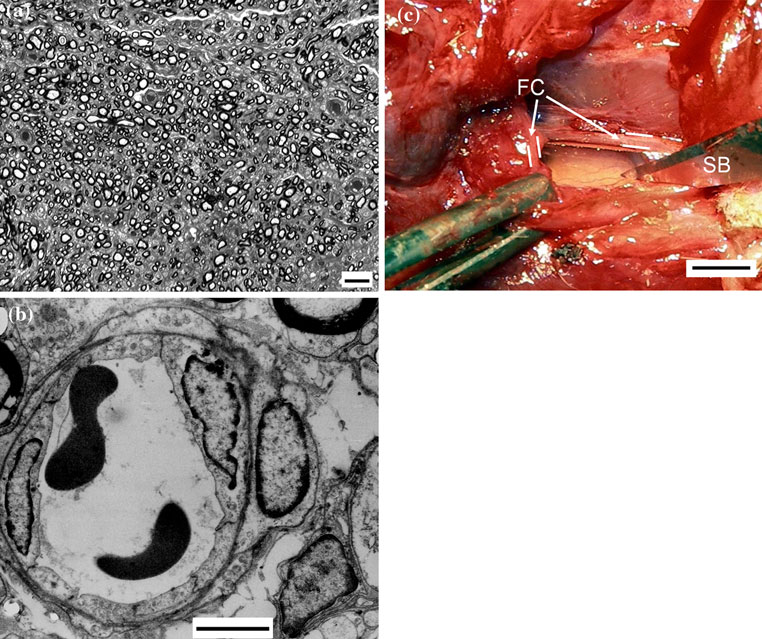
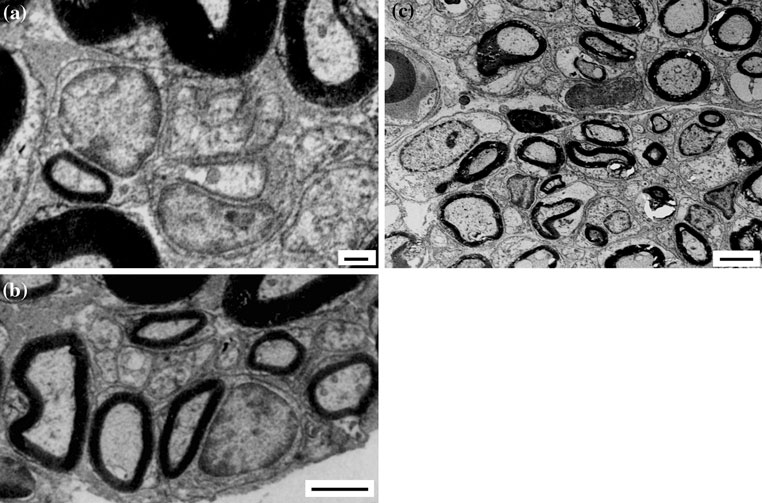
Fig. 4 a A transverse section of the nerve regenerated inside the
a sharp blade (SB) and the transparent wall of the regeneration
NeuroBox (toluidine blue; bar = 10 lm). b A tiny intraneural
chamber showed tiny blood vessels in the regenerated nerve
capillary in the regenerated nerve (TEM; bar = 1 lm). c A thick
fibrous capsule (FC) was found around the nerve-guide; it was cut by
vascularization. TEM showed smaller fibers, with thin
transection with negligible or absent gap, prescribes the
myelin sheath, which represent newly regenerating axons;
joining of the two nerve-stumps by an end-to-end suture
there are also larger fibers with disruption myelin figures,
(neurorraphy) ] and it is widely accepted that the stumps
as expected in degenerating axons proximal to a lesion
must not be sutured under tensional stress ] otherwise
(Fig. b). In DGR, tiny myelinated and non myelinated
the development of a fibroblastic and myofibroblastic
fibers represent axons that entered the distal stump; there
proliferation will be greatly favoured. The latter phenom-
is, also, the presence of Schwann's cells digesting the
enon will impair and eventually stop any axonal regener-
myelin of axons which underwent Wallerian degeneration
ation []. However the use of stitches (both degradable or
not degradable) represent a significative local inflammatory
On the contrary, no regeneration was observed in G2,
stimulus [, ] even without tensioning the stumps and is
where the gap was left un-treated (3 after 1 month; 4 after
able to provoke enough fibrosis to impair nerve regener-
2 months; as shown in Table ).
One should not assume the fibroblastic and myofibrob-
lastic proliferation to be un-avoidable, just because of our
present inability to abandon the use of stitches. Researchon ‘‘stitch-less'' techniques in any surgery associated with
There are several proposals in the literature about the best
artificial nerve guides seems instrumental, in our opinion,
solution to adopt for each of the three districts which
to the successful development of the guides themselves and
constitute an artificial nerve guide [but the suture dis-
glues seem to most straightforward option.
trict, in our opinion, has received minor attention until
The present knowledge on nerve-glues is, however,
now. This district is the mechanical interface between the
limited. In the past, cyano-acrylic glues, and ethyl-2-
guide and the nerve and is a crucial point both biologically
cyanoacrylate in particular, have been associated with
and surgically; established surgical treatment, in acute
Asthma [] and Allergic Contact Dermatitis –
J Mater Sci: Mater Med (2010) 21:1979–1987
Author's personal copy
Fig. 5 A clear fascicular structure of the nerve was preserved in the
of glue were found (a and b: toluidine blue; bar = 0.1 mm) (c and d:
proximal glueing region (a, c) and in the distal glueing region (b, d),
H&E; bar = 10 lm)
were well demarcated from the surrounding epineurium; no particles
Fig. 6 a Transmission electron microscopy of the proximal glueing
to a lesion (bar = 10 lm). c The distal glueing region shows tiny
region with myelinated and non-myelinated fibers (bar = 1 lm)
myelinated and non myelinated fibers as well; Schwann's cells are
representing newly regenerating axons; b there are also fibers with
digesting the myelin of axons which underwent Wallerian degener-
disruption myelin figures, as expected in degenerating axons proximal
ation (bar = 10 lm)
J Mater Sci: Mater Med (2010) 21:1979–1987
Author's personal copy
Furthermore, ethyl-2-cyanoacrylate has been correlated
Discussing the experimental design, it must be said that
with neuropathy in sporadic cases in which, it must be
the limited number of implanted guides was dictated
noted, other factors were possibly involved and great
mostly by the high costs associated with their production,
quantity and/or prolonged exposure were reported [–
while the short length of the treated gap was chosen to
As a group of rapidly polymerizing adhesives, cyano-
simplify the experiment aimed primarily at testing the
acrylates have found surgical applications as skin-wound
adequacy of the new concept of the ‘‘stitch-less'' guide.
sutures as well as hemostatic and embolizing agents [
Despite these limitations, a safe use of ethyl-2-cyanoac-
In more recent literature promising results have been
rylate in direct contact with a nerve regenerating inside the
reported with cyanoacrylate molecules in nerve surgery, in
NeuroBox was demonstrated and this maintains our com-
mitment to refine a ‘‘stich-less'' surgical technique for
2-octyl-cyanoacrylate. These papers show that cyanoacrylic
nerve repair. Any progress in nerve-guide surgery will lead,
glues can be used in direct contact with the nerve –].
someday, to a significant reduction in nerve-autografts
The actual use of cyanoacrylate glue in a true surgical
requirement; this will mean lesser associated morbidity,
setting may bear a lot of technical difficulties and this may
shorter surgical time, minor complexity. In brief, a real
explain, in part, early negative recommendations
improvement for treating a large number of patients.
First: it is not easy to control the curing time of the glue,which should be not too fast, so giving the surgeon ade-
The NeuroBox is an international patent of the
Catholic University in Rome (WO/2008/029373).
quate time to accurately put the stumps in place, but (at thesame time) should be not too slow, to avoid the accidentalflow of part of the glue in front of the nerve stump(bringing the misfortunate consequence of sealing it and
impairing the regeneration process). Second: to find asuitable method of delivering the glue in a real surgical
1. Sinis N, Schaller HE, Schulte-Eversum C, Lanaras T, Schlosshauer
environment may be an additional problem; for example,
B, Doser M, et al. Comparative neuro tissue engineering using
tiny quantities required by a digital nerve suture could
different nerve guide implants. Acta Neurochir Suppl. 2007;
polymerize early inside a microscopic delivery tube, before
2. Rappaport WD, Valente J, Hunter G. Clinical utilization and
reaching the stump. Third: in a real surgical setting, an
complications of sural nerve biopsy. Am J Surg. 1993;166:252–6.
unpredictable bleeding may vanish the effectiveness of a
3. Taras JS, Nanavati V, Steelman P. Nerve conduits. J Hand Ther.
glueing procedure; this could be a quite frequent occur-
rence if nerve surgery has to be performed with a limited
4. Bora FW Jr. Peripheral nerve repair in cats. The fascicular stitch.
J Bone Joint Surg Am. 1967;49:659–66.
(or absent) intraoperative ischemia (tourniquet), as advo-
5. Dahlin LB. Nerve injuries. Curr Orthop. 2008;22:9–16.
cated to better preserve the vitality of the nerve.
6. Neubauer D, Graham JB, Muir D. Chondroitinase treatment
In the present work, Authors found that ethyl-2-cyano-
increases the effective length of acellular nerve grafts. Exp
acrylate was easily applied in the peculiar construct of the
7. Ijkema-Paassen J, Jansen K, Gramsbergen A, Meek MF. Tran-
NeuroBox and nerve regeneration was not affected by the
section of peripheral nerves, bridging strategies and effect eval-
presence of the acrylic glue all around the epineural sheath
uation. Biomaterials. 2004;25(9):1583–92.
of the glueing regions. In particular, no alterations were
8. Sinis N, Schaller HE, Schulte-Eversum C, Schlosshauer B, Doser
found in the morphology of axons and Schwann's cells and
M, Dietz K, et al. Nerve regeneration across a 2-cm gap in the ratmedian nerve using a resorbable nerve conduit filled with
these results confirm that ethyl-2-cyanoacrylate can be
Schwann cells. J Neurosurg. 2005;103(6):1067–76.
used in direct contact with the nerve [
9. Schlosshauer B, Dreesmann L, Schaller HE, Sinis N. Synthetic
The peculiar geometry of the NeuroBox, in our opinion,
nerve guide implants in humans: a comprehensive survey. Neu-
helps greatly in the surgical application of the glue and
10. Meek MF, Coert JH. US Food and Drug Administration/
greatly reduces the quantity which is needed; this may, in
Conformit Europe-approved absorbable nerve conduits for clin-
turn, minimize the fibroblastic response. However, the
ical repair of peripheral and cranial nerves. Ann Plast Surg. 2008;
device may possibly have a further role in diverging the
fibroblasts away from the Regeneration Chamber since it
11. Ahmed MR, Vairamuthu S, Shafiuzama M, Basha SH, Jayakumar
R. Microwave irradiated collagen tubes as a better matrix for
was observed that most of the fibroblasts in the area are
peripheral nerve regeneration. Brain Res. 2005;1046(1–2):55–67.
engaged in the formation of an outside capsule around the
12. Bertleff MJ, Meek MF, Nicolai JP. A prospective clinical eval-
PMMA-made NeuroBox. It may be speculated that the
uation of biodegradable neurolac nerve guides for sensory nerve
device acts as a decoy for the large number of fibroblasts
repair in the hand. J Hand Surg [Am]. 2005;30(3):513–8.
13. Bozkurt A, Brook GA, Moellers S, Lassner F, Sellhaus B, Weis J,
that, instead of entangling the tiny regenerating axons
et al. In vitro assessment of axonal growth using dorsal root
inside the Regeneration Chamber, primarily attack the
ganglia explants in a novel three-dimensional collagen matrix.
nerve-guide outer structure.
Tissue Eng. 2007;13(12):2971–9.
J Mater Sci: Mater Med (2010) 21:1979–1987
Author's personal copy
14. Bunting S, Di Silvio L, Deb S, Hall S. Bioresorbable glass fibres
31. Phillips JB, Bunting SC, Hall SM, Brown RA. Neural tissue
facilitate peripheral nerve regeneration. J Hand Surg [Br]. 2005;
engineering: a self-organizing collagen guidance conduit. Tissue
15. Chang CJ, Hsu SH, Yen HJ, Chang H, Hsu SK. Effects of
32. Scherman P, Kanje M, Dahlin LB. Sutures as longitudinal guides
unidirectional permeability in asymmetric poly(DL-lactic acid-
for the repair of nerve defects—influence of suture numbers and
co-glycolic acid) conduits on peripheral nerve regeneration:
reconstruction of nerve bifurcations. Restor Neurol Neurosci.
an in vitro and in vivo study. J Biomed Mater Res B Appl Bio-
33. Stokols S, Tuszynski MH. Freeze-dried agarose scaffolds with
16. Chang JY, Lin JH, Yao CH, Chen JH, Lai TY, Chen YS. In vivo
uniaxial channels stimulate and guide linear axonal growth fol-
evaluation of a biodegradable EDC/NHS-cross-linked gelatin
lowing spinal cord injury. Biomaterials. 2006;27(3):443–51.
peripheral nerve guide conduit material. Macromol Biosci. 2007;
34. Stokols S, Sakamoto J, Breckon C, Holt T, Weiss J, Tuszynski
MH. Templated agarose scaffolds support linear axonal regen-
17. Chavez-Delgado ME, Gomez-Pinedo U, Feria-Velasco A,
eration. Tissue Eng. 2006;12(10):2777–87.
Huerta-Viera M, Castaneda SC, Toral FA, et al. Ultrastructural
35. Sundback CA, Shyu JY, Wang Y, Faquin WC, Langer RS,
analysis of guided nerve regeneration using progesterone- and
Vacanti JP, et al. Biocompatibility analysis of poly(glycerol
pregnenolone-loaded chitosan prostheses. J Biomed Mater Res B
sebacate) as a nerve guide material. Biomaterials. 2005;26(27):
Appl Biomater. 2005;74(1):589–600.
18. Chen YS, Chang JY, Cheng CY, Tsai FJ, Yao CH, Liu BS. An
36. Tos P, Battiston B, Nicolino S, Raimondo S, Fornaro M, Lee JM,
in vivo evaluation of a biodegradable genipin-cross-linked gela-
et al. Comparison of fresh and predegenerated muscle-vein-
tin peripheral nerve guide conduit material. Biomaterials. 2005;
combined guides for the repair of rat median nerve. Microsur-
19. Huang YC, Huang YY, Huang CC, Liu HC. Manufacture of
37. Tyner TR, Parks N, Faria S, Simons M, Stapp B, Curtis B, et al.
porous polymer nerve conduits through a lyophilizing and wire-
Effects of collagen nerve guide on neuroma formation and neu-
heating process. J Biomed Mater Res B Appl Biomater. 2005;
ropathic pain in a rat model. Am J Surg. 2007;193(1):e1–6.
38. Uebersax L, Mattotti M, Papaloizos M, Merkle HP, Gander B,
20. Inada Y, Hosoi H, Yamashita A, Morimoto S, Tatsumi H,
Meinel L. Silk fibroin matrices for the controlled release of nerve
Notazawa S, et al. Regeneration of peripheral motor nerve gaps
growth factor (NGF). Biomaterials. 2007;28(30):4449–60.
with a polyglycolic acid-collagen tube: technical case report.
39. Yoshitani M, Fukuda S, Itoi S, Morino S, Tao H, Nakada A, et al.
Experimental repair of phrenic nerve using a polyglycolic acid and
21. Jansen K, Meek MF, van der Werff JF, van Wachem PB, van
collagen tube. J Thorac Cardiovasc Surg. 2007;133(3):726–32.
Luyn MJ. Long-term regeneration of the rat sciatic nerve through
40. Wieken K, Angioi-Duprez K, Lim A, Marchal L, Merle M. Nerve
anastomosis with glue: comparative histologic study of fibrin and
guide: tissue reactions with focus on collagen III/IV reformation.
cyanoacrylate glue. J Reconstr Microsurg. 2003;19(1):17–20.
J Biomed Mater Res A. 2006;69(2):334–41.
41. Klein SM, Nielsen KC, Buckenmaier CC III, Kamal AS, Rubin
22. Lietz M, Ullrich A, Schulte-Eversum C, Oberhoffner S, Fricke C,
Y, Steele SM. 2-Octyl cyanoacrylate glue for the fixation of
Muller HW, et al. Physical and biological performance of a novel
continuous peripheral nerve catheters. Anesthesiology. 2003;98(2):
block copolymer nerve guide. Biotechnol Bioeng. 2006;93(1):
42. Choi BH, Kim BY, Huh JY, Lee SH, Zhu SJ, Jung JH, et al.
23. Lietz M, Dreesmann L, Hoss M, Oberhoffner S, Schlosshauer B.
Microneural anastomosis using cyanoacrylate adhesives. Int J
Neuro tissue engineering of glial nerve guides and the impact of
Oral Maxillofac Surg. 2004;33(8):777–80.
different cell types. Biomaterials. 2006;27(8):1425–36.
43. Pin˜eros-Ferna´ndez A, Rodeheaver PF, Rodeheaver GT. Octyl
24. Madaghiele M, Sannino A, Yannas IV, Spector M. Collagen-
2-cyanoacrylate for repair of peripheral nerve. Ann Plast Surg.
based matrices with axially oriented pores. J Biomed Mater Res
44. Landegren T, Risling M, Brage A, Persson JK. Long-term results
25. Marchesi C, Pluderi M, Colleoni F, Belicchi M, Meregalli M,
of peripheral nerve repair: a comparison of nerve anastomosis
Farini A, et al. Skin-derived stem cells transplanted into resorb-
with ethyl-cyanoacrylate and epineural sutures. Scand J Plast
able guides provide functional nerve regeneration after sciatic
Reconstr Surg Hand Surg. 2006;40(2):65–72.
nerve resection. Glia. 2007;55(4):425–38.
45. Landegren T, Risling M, Persson JK. Local tissue reactions after
26. Merolli A, Rocchi L, Catalano F, Planell J, Engel E, Martı´nez
nerve repair with ethyl-cyanoacrylate compared with epineural
E, et al. In vivo regeneration of rat sciatic nerve in a double-
sutures. Scand J Plast Reconstr Surg Hand Surg. 2007;41(5):
halved stitch-less guide: a pilot-study. Microsurgery. 2009;29(4):
46. Elgazzar RF, Abdulmajeed I, Mutabbakani M. Cyanoacrylate
27. Oh SH, Lee JH. Fabrication and characterization of hydrophilized
glue versus suture in peripheral nerve reanastomosis. Oral
porous PLGA nerve guide conduits by a modified immersion
Surg Oral Med Oral Pathol Oral Radiol Endod. 2007;104(4):
precipitation method. J Biomed Mater Res A. 2007;80(3):530–8.
28. Patel M, Vandevord PJ, Matthew H, Wu B, DeSilva S, Wooley
47. Rickett T, Li J, Patel M, Sun W, Leung G, Shi R. Ethyl-cyano-
PH. Video-gait analysis of functional recovery of nerve repaired
acrylate is acutely nontoxic and provides sufficient bond strength
with chitosan nerve guides. Tissue Eng. 2006;12(11):3189–99.
for anastomosis of peripheral nerves. J Biomed Mater Res A.
29. Patel M, Mao L, Wu B, Vandevord PJ. GDNF-chitosan blended
nerve guides: a functional study. J Tissue Eng Regen Med. 2007;
48. Li J, Yan JG, Ai X, Hu S, Gu YD, Matloub HS, et al. Ultra-
structural analysis of peripheral-nerve regeneration within a nerve
30. Pereira Lopes FR, Camargo de Moura Campos L, Dias Correˆa J
conduit. J Reconstr Microsurg. 2004;20(7):565–9.
Jr, Balduino A, Lora S, Langone F, et al. Bone marrow stromal
49. Merolli A, Rocchi L. Peripheral nerve regeneration by artificial
cells and resorbable collagen guidance tubes enhance sciatic
nerve guides. In: Merolli A, Joyce TJ, editors. Biomaterials in
nerve regeneration in mice. Exp Neurol. 2006;198(2):457–68.
hand surgery. Heidelberg: Springer; 2009. p. 127–43.
J Mater Sci: Mater Med (2010) 21:1979–1987
Author's personal copy
50. Millesi H. Reappraisal of nerve repair. Surg Clin North Am.
58. Chan CC, Cheong TH, Lee HS, Wang YT, Poh SC. Case of
occupational asthma due to glue containing cyanoacrylate. Ann
51. Millesi H, Meissl G, Berger A. Further experience with inter-
Acad Med Singap. 1994;23:731–3.
fascicular grafting of the median, ulnar and radial nerves. J Bone
59. Hanft JR, Kashuk KB, Toney ME, McDonald TD. Peripheral
Joint Surg Am. 1976;58(2):209–18.
neuropathy as a result of cyanoacrylate toxicity. J Am Pediatr
52. Yannas IV, Zhang M, Spilker MH. Standardized criterion to
Med Assoc. 1991;12:653–5.
analyze and directly compare various materials and models for
60. Page EH, Pajeau AK, Arnold TC, Fincher AR, Goddard MJ.
peripheral nerve regeneration. J Biomater Sci Polym Ed. 2007;
Peripheral neuropathy in workers exposed to nitromethane. Am J
Ind Med. 2001;40(1):107–13.
53. Belsito DV. Contact dermatitis to ethyl-cyanoacrylate-containing
61. Kuroki T, Aoki K, Aoki Y, Nemoto A, Yamazaki T, Katsume M,
glue. Contact Dermat. 1987;17(4):234–6.
et al. Cranial nerve pareses following wrapping of a ruptured
54. Bruze M, Bjorkner BM, Lepoittvin JP. Occupational allergic
dissecting vertebral artery aneurysm: a possible complication of
contact dermatitis from ethyl cyanoacrylate. Contact Dermat.
cyanoacrylate glue—case report. Neurol Med Chir. 2003;43(1):
55. Conde-Salazar L, Rojo S, Guimaraens D. Occupational contact
62. Vinters HV, Galil KA, Lundie MJ, Kaufmann JC. The histotox-
dermatitis from cyanoacrylates. Am J Contact Dermat. 1998;9(3):
icity of cyanoacrylates. A selective review. Neuroradiology. 1985;
56. Nakazawa T. Occupational asthma due to alkyl cyanoacrylate.
J Occup Med. 1990;32(8):709–10.
57. Savonius B, Keskin H, Tuppurainen M, Kanerva L. Occupational
respiratory disease caused by acrylates. Clin Exp Allergy. 1993;23:416–24.
Source: http://www.chirurgiadellamanoroma.it/pdf/235_-_in_vivo_study_of_ethyl-2-cyanoacrylate_appli.pdf
32_RODRIGUEZ MARADIAGA_pp_484-495.QXD_Layout 1 07/02/13 16:06 Pagina 484 The Global Quest for Tranquillitas Ordinis. Pacem in Terris, Fifty Years Later Pontifical Academy of Social Sciences, Acta 18, 2013 www.pass.va/content/dam/scienzesociali/pdf/acta18/acta18-rodriguezmaradiaga.pdf Aproximación geopolítica a la justiciaen un mundo globalizado
Researchmaster Infection & Immuntiy Laboratory rotations & Reaearch topics IMMUNOLOGY – LABROTATIONS & RESEARCH TOPICS Researchmaster Infection & Immuntiy Laboratory rotations & Reaearch topics Title: (Immuno)pathogenesis of chronic lymphocytic leukemia Workgroupleader: dr. A.W. Langerak T: 010-704 4089 E: [email protected] W: http://www.erasmusmc.nl/immunologie/onderzoek/moleculaireimmunologie/mid/?lang=en Background Chronic lymphocytic leukemia (CLL) is the most frequent type of leukemic proliferation in the Western world. CLL is found in adults and typically associated with age. The majority of CLL cases is of B-cell type, while a minority derives from T lymphocytes (also called T-cell large granular lymphocyte leukemia, T-LGL). Over the last years it has become increasingly clear that CLL is a heterogeneous disease, with a variable clinical course and differences in survival. CLL is an example of a multi-factorial disease, in which both genetic and micro-environmental factors contribute to leukemogenesis. Although in recent years many studies have focused on prognostic markers, there is still no complete picture of the factors that are involved in the (immuno)pathogenesis and that are determining for the prognosis.








